Pill Connect
Don't let unmeasured adherence undermine your trial.
Pill Connect captures every dose at the bottle, sending it automatically to a monitoring system. If an adherence issue is seen, care teams receive automated alerts highlighting the issue. This way, everyone can get on with their job knowing that adherence is being measured and accounted for.

How it works
A feedback loop that runs in the background.
Pill Connect closes the loop between a patient deviating from the protocol and a site team knowing, without adding a task to anyone's day.
-
The patient
Patients just take their medicine.
Each pill is dispensed with a button press. This makes data capture and recording automatic and means the time-stamp represents the live event of the patient dispensing and is confirmed by sensors watching the pill eject.
Explore the dispenser → -
The data
The data moves on its own.
It leaves the bottle automatically so there's nothing for the patient to open or manually sync with. Remote, continuous per-pill, real-time data on exactly how patients are dispensing, without asking them to do anything beyond dispense the pill.
-
The site team
Your site team just supports patients.
When dosing slips, an email reaches the team that needs this information, with exactly what happened and when. No dashboards to police, just an alert to reach out when it matters.
See real-time monitoring →
The result The most accurate picture of what's actually happening, turned into personalised support exactly when it's needed.
Adherence you can't see is risk you can't price.
If adherence isn't measured, you can't see it slipping and a working drug with poor adherence can look just like one that failed. Objective, per-pill data keeps your efficacy signal clean.
Why real-time matters →Watch
Digital drug accountability.
See how Pill Connect turns every dispense into an objective, auditable record.
Latest
News & Updates

29 May 2026
Automated, real-time adherence monitoring
New feature: automated data reading from a smart pill dispenser
21 May 2026
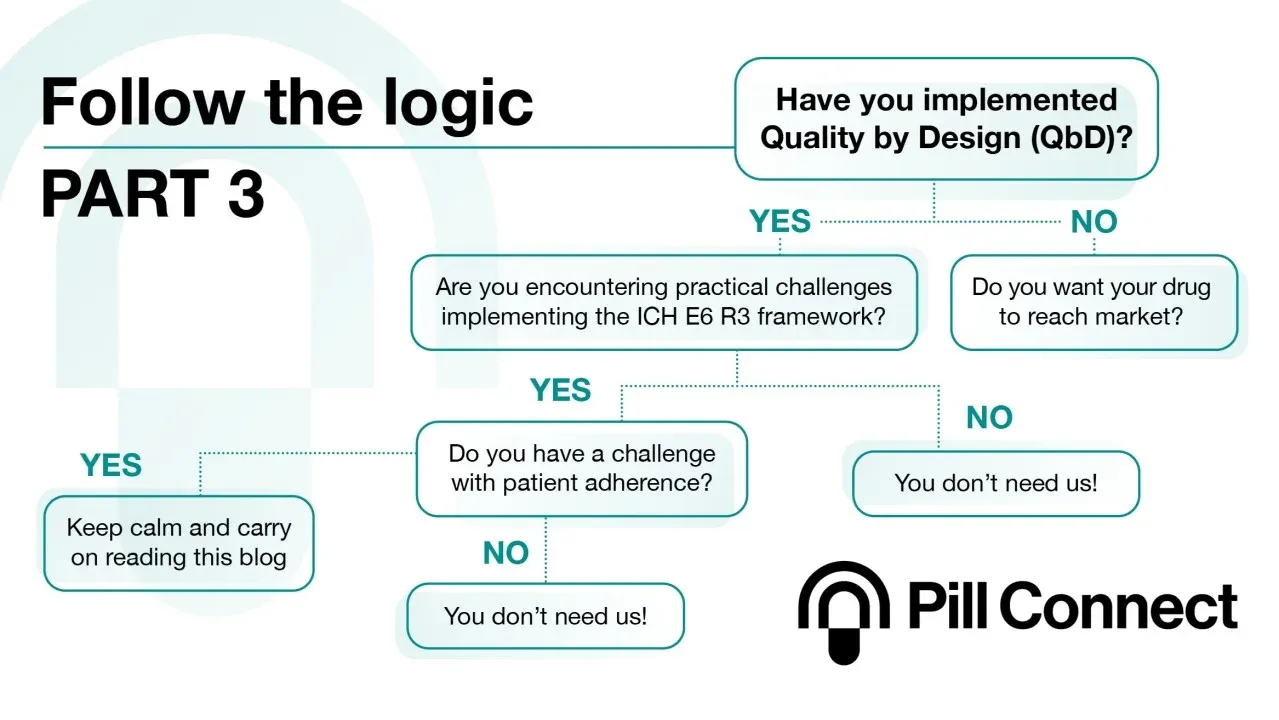
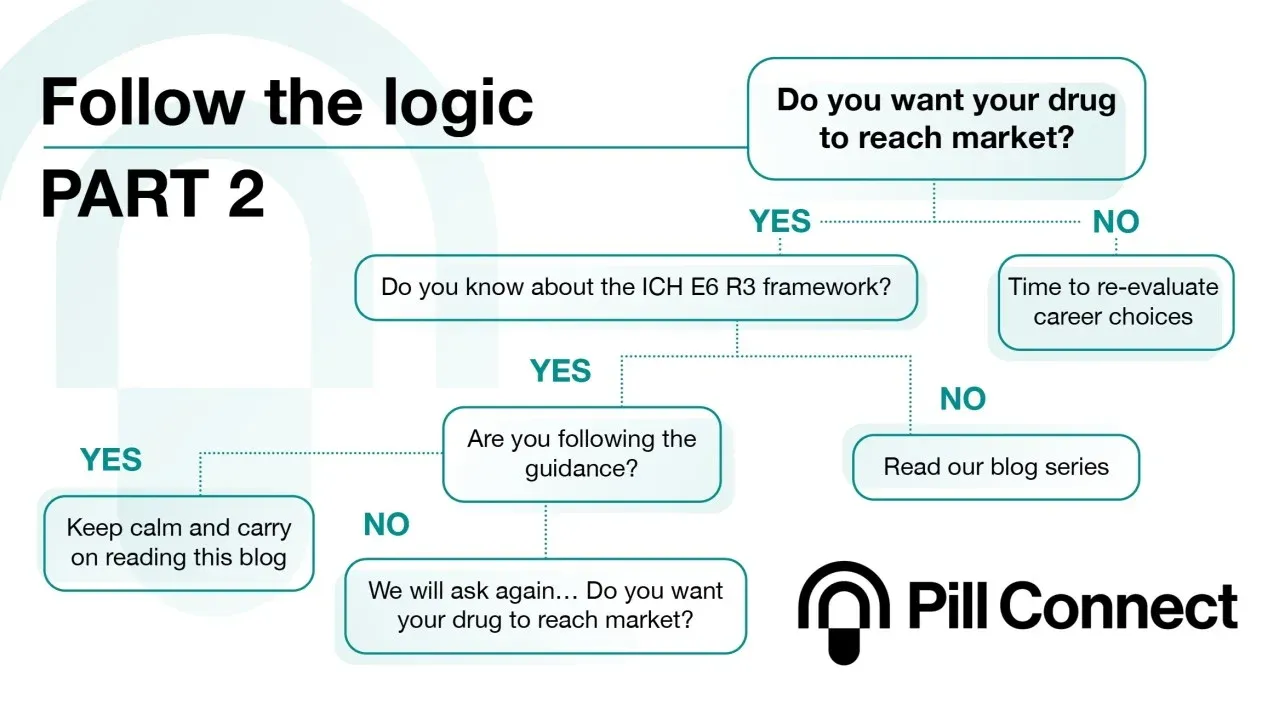
Protecing quality in clinical trials
Real-time, pill-level adherence monitoring to de-risk rare disease trials and meet ICH E6(R3). Read the full story.
14 May 2026
Why regulators are turning the screw on adherence in clinical trials
ICH E6(R3) sharpens the focus on critical-to-quality. Stronger adherence controls are a way sponsors meet it.
.ChOuMaJr_vFJoJ.webp)
7 May 2026
Highlighting the real Achilles Heel of rare disease trials
Retention is the Achilles heel of rare disease trials. How could early detection of dropouts help outcomes and where does this fit ICH E6(R3)?

24 February 2026
What a technical evaluation taught us about adherence as a behaviour
The behavioural patterns observed in this technical evaluation are worth sharing about how adherence is monitored in clinical trials.

11 February 2026
The AI Adherence Trap: Better Data, Not Fewer Humans
A blog on: The adherence data quality problem, The risks of AI-driven coaching, What responsible AI actually looks like
Behind the product
Our Team
Joined Elucid mHealth in 2013 as CTO. Moved to CEO and rebranded to Pill Connect in 2017. Lead the original technical design of the companies connected devices. Inventor on several patents and now leads customer and partner relationships and clinical trials. Previously completed PhD in Machine Learning and Pattern Recognition.
Leads technical development and quality. Named inventor on several patents. Twelve years in regulated connected products. Previously Lead Systems Engineer at GE Healthcare, following its acquisition of Monica Healthcare.
Highly experienced Pharma device expert and company Chairman and NED. Experience includes Aptar Pharma, Bespak and MWV. Now works with NanoPharm, LTS, Merxin and Becton Dickinson.
Founder of Synexus, the global leader in clinical trial recruitment. Then founded Panthera Biopartners, a new recruitment company which he exited again. Led both companies as CMO. NHS GP.
Founder and CEO of Monica Healthcare, a GE company. Now working as NED and advisor to medical device start ups.
Chartered accountant, previously worked at Astra Zeneca and Advanced Medical Systems.
FAQ
Questions & Answers.
Everything sponsors, CROs and research sites typically ask during evaluation.
Can't find what you're looking for? Contact us →
Clinical Use & Study Design
-
Is Pill Connect suitable for use in clinical trials and launched drug programs?
Yes. Pill Connect is suitable for use in clinical trials as well as launched drug programs where objective medication adherence data is required for patients or participants dosing at home.
-
Is Pill Connect suitable for decentralized or hybrid clinical trials?
Yes. Pill Connect supports decentralized and hybrid clinical trials by enabling medication adherence data to be collected remotely, without requiring participants to attend site visits solely for adherence monitoring.
-
How does Pill Connect compare to pill counts or patient diaries?
Pill Connect provides objective, time-stamped dispensing data, reducing reliance on indirect adherence measures such as pill counts or self-reported patient diaries, which can be incomplete or inaccurate.
-
What adherence data does Pill Connect collect in a clinical trial?
Pill Connect records dispensing events, including the date and time each individual pill is dispensed, as well as bottle swap events. This data can be used to support medication adherence analysis during a clinical trial.
Discuss your trial.
Interested in using the Pill Connect Smart Dispenser in your trial — or want to learn more about how it can be used? Get in touch.
- Email info@pill-connect.com
- LinkedIn pillconnect

